Synedeica Somatotropin HGH 120IU Injection Pen Kit
£130.00
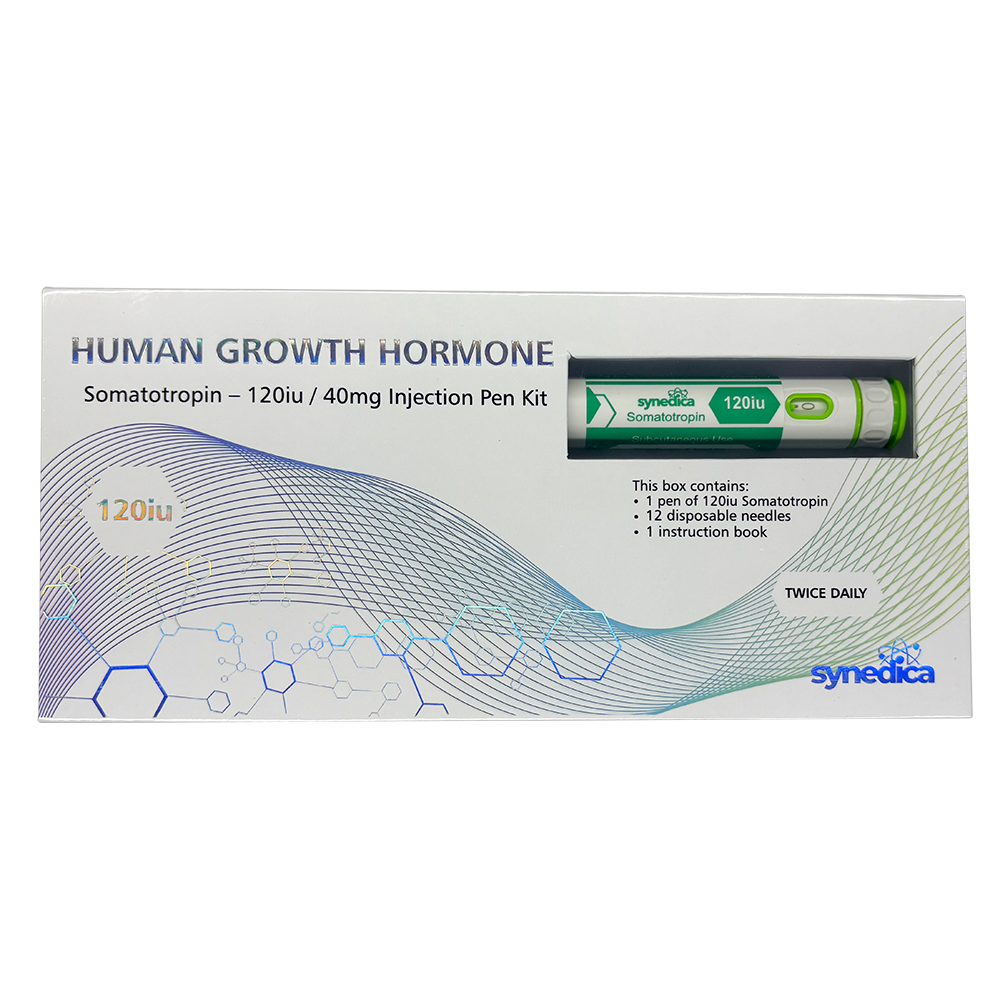
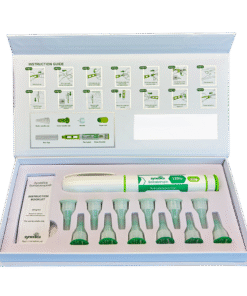
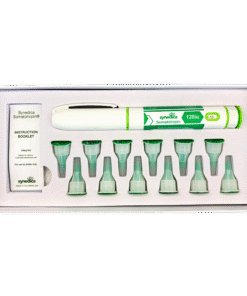
Synedica Somatotropin HGH 120IU Injection Pen Kit
The Synedica Somatotropin HGH Injection Pen Kit is a premium recombinant human growth hormone (HGH) developed for advanced laboratory and pharmaceutical research across the UK and European markets. Also known as Somatotropin, this synthetic peptide is widely studied in endocrinology, metabolism, and cell growth research due to its role in regulating growth hormone pathways and protein synthesis.
Synedica Somatotropin HGH 120IU Injection Pen Kit
The Synedica Somatotropin HGH Injection Pen Kit is a premium recombinant human growth hormone (HGH) developed for advanced laboratory and pharmaceutical research across the UK and European markets. Also known as Somatotropin, this synthetic peptide is widely studied in endocrinology, metabolism, and cell growth research due to its role in regulating growth hormone pathways and protein synthesis.
Somatotropin is a key compound in scientific investigations exploring muscle development, recovery processes, and fat metabolism at a biochemical level. Its interaction with growth hormone receptors makes it particularly valuable in controlled studies focused on anabolic signalling, tissue regeneration, and metabolic function.
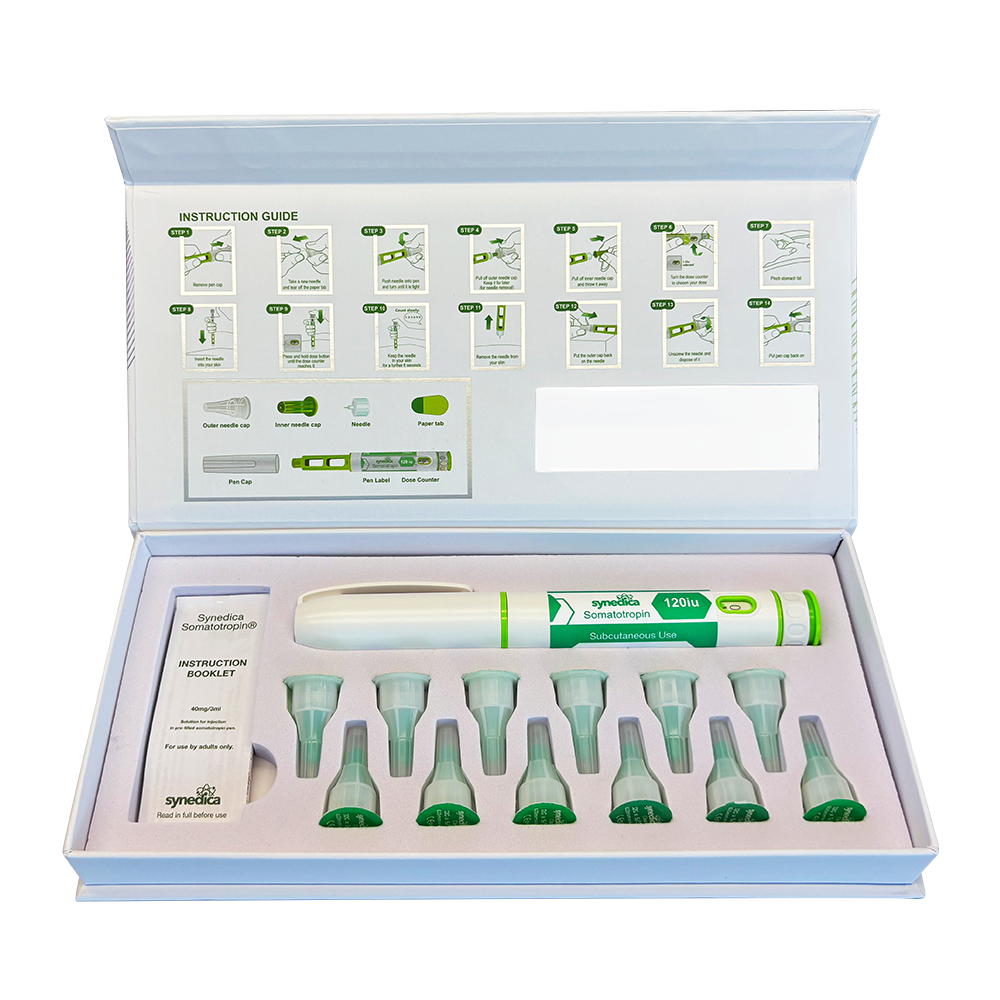
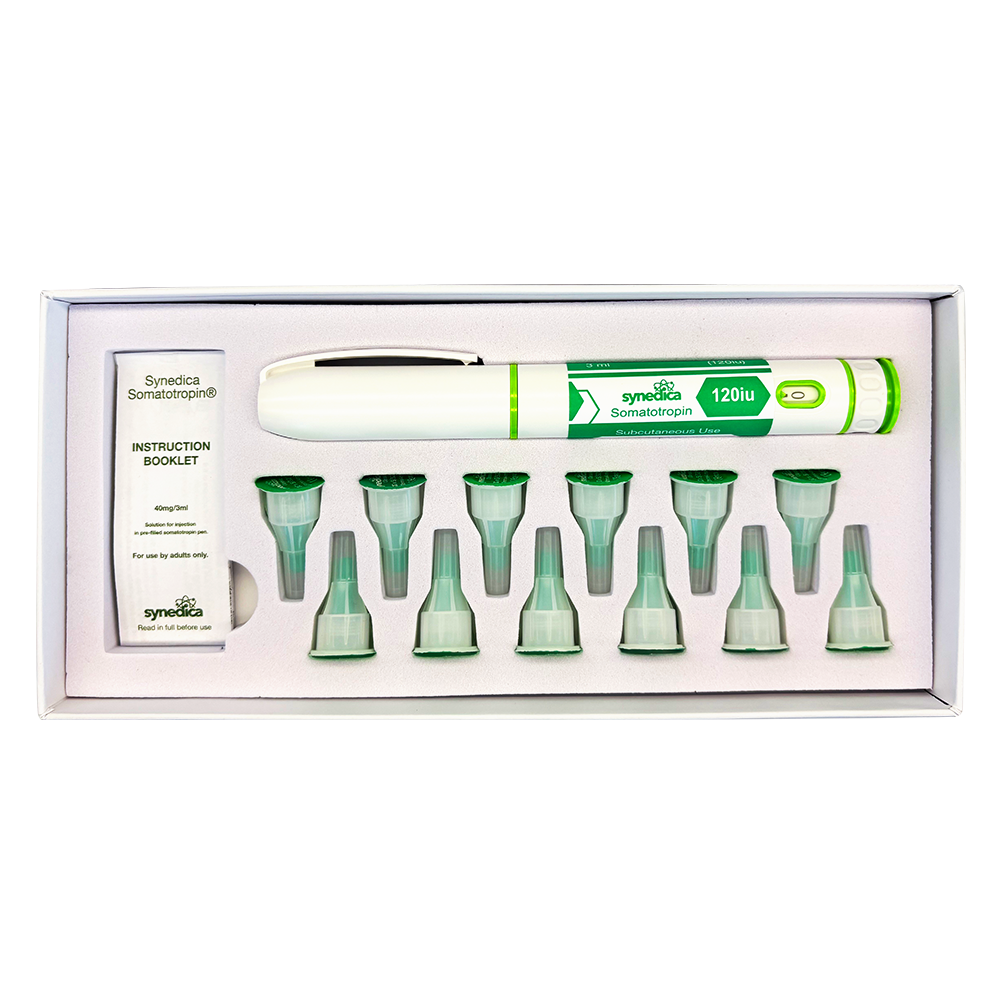
The Synedica Somatotropin kit is manufactured to high purity standards and supplied in a format designed for precision handling in laboratory environments. The 120IU injection pen kit is developed for controlled dosing accuracy and consistency, supporting repeatable research outcomes in both academic and commercial settings.
This product is typically supplied as part of a complete research kit, which may include multiple vials of lyophilised somatotropin alongside bacteriostatic water for reconstitution. The formulation is designed for subcutaneous delivery within laboratory protocols, enabling researchers to study peptide stability, absorption pathways, and delivery mechanisms under controlled conditions.
Within research settings, somatotropin is often examined in dose-response studies to better understand its effects on cellular growth, metabolic regulation, and protein turnover. Any dosing frameworks referenced in scientific literature are strictly for experimental design purposes and must only be applied within regulated laboratory environments by qualified professionals.
Research & Development
Synedica Somatotropin is widely used in stability testing, where it is analysed under thermal, enzymatic, and oxidative conditions to determine degradation patterns, shelf-life, and formulation resilience. It also plays an important role in formulation development, supporting research into improved solubility, enhanced bioavailability, and structural integrity of peptide-based compounds.
In excipient compatibility studies, somatotropin is evaluated alongside inactive ingredients to ensure formulation safety, consistency, and performance. Protease resistance testing allows researchers to assess how the peptide responds to enzymatic breakdown, contributing to the development of more stable and durable compounds.
Advanced applications include encapsulation and delivery system research, where somatotropin is incorporated into liposomes, microspheres, and other carrier technologies designed to protect the peptide and enable controlled release. It is also used in administration route assessments, supporting research into injectable delivery systems, dermal absorption models, and in vitro laboratory testing.
This product is supplied strictly for research and development purposes only. It is not approved for human or veterinary use.
Be the first to review “Synedeica Somatotropin HGH 120IU Injection Pen Kit” Cancel reply
Related products
Peptide
Peptide
Peptide
Peptide







Reviews
There are no reviews yet.