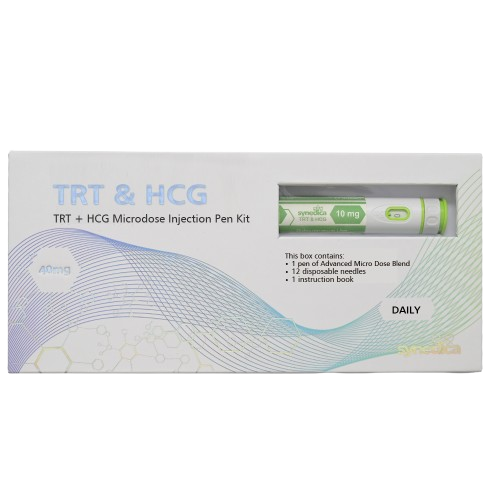
Synedica TRT + HCG Injection Pen Kit
£100.00
Synedica TRT + HCG Injection Pen Kit
The Synedica TRT + HCG Daily Injection Pen Kit is a research-grade hormonal combination kit developed for advanced laboratory and pharmaceutical research across the UK and European markets. This formulation combines Testosterone Cypionate with Human Chorionic Gonadotropin, two compounds widely studied in endocrinology for their roles in hormone regulation, reproductive biology, and metabolic function.
This kit is designed to support controlled investigation into testosterone delivery models and hormonal balance mechanisms. Testosterone cypionate is commonly researched for its long-acting androgenic properties, while HCG is studied for its ability to stimulate endogenous hormone pathways. Together, they are frequently examined in research exploring hormonal stability, endocrine signalling, and physiological response patterns.
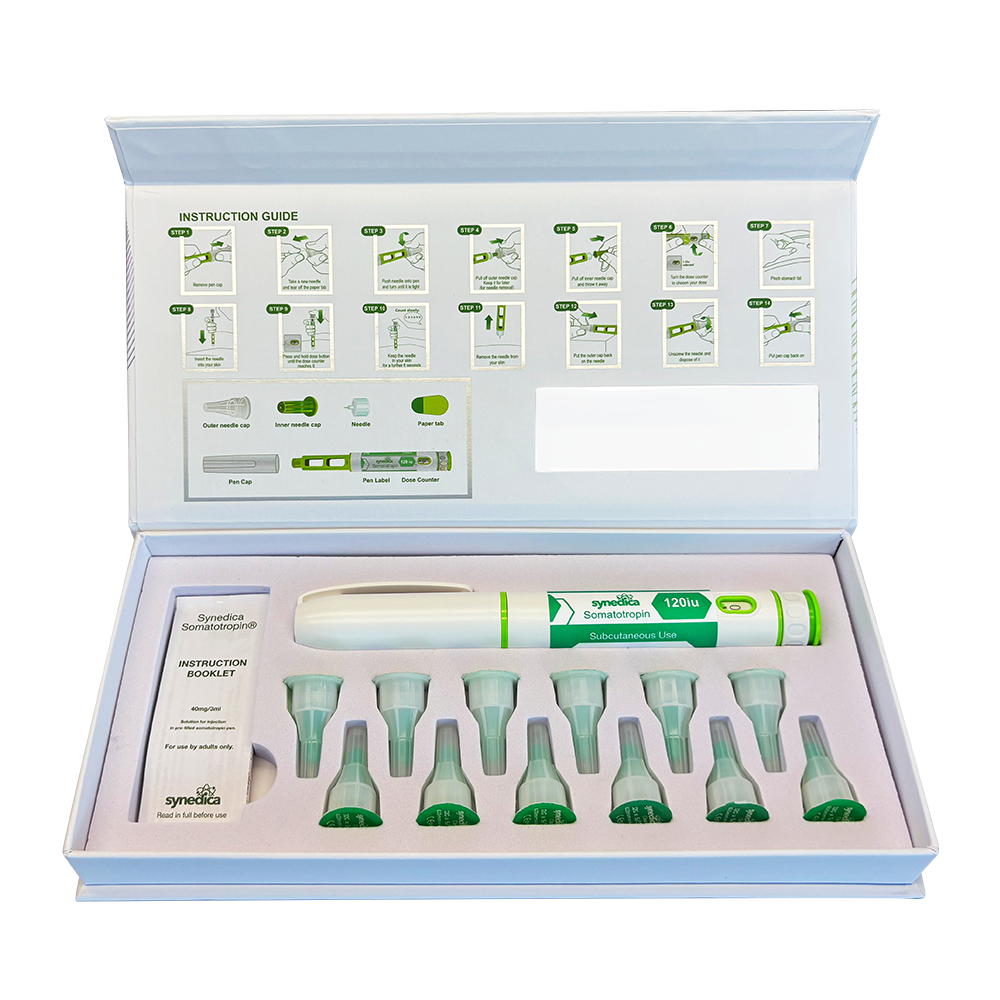
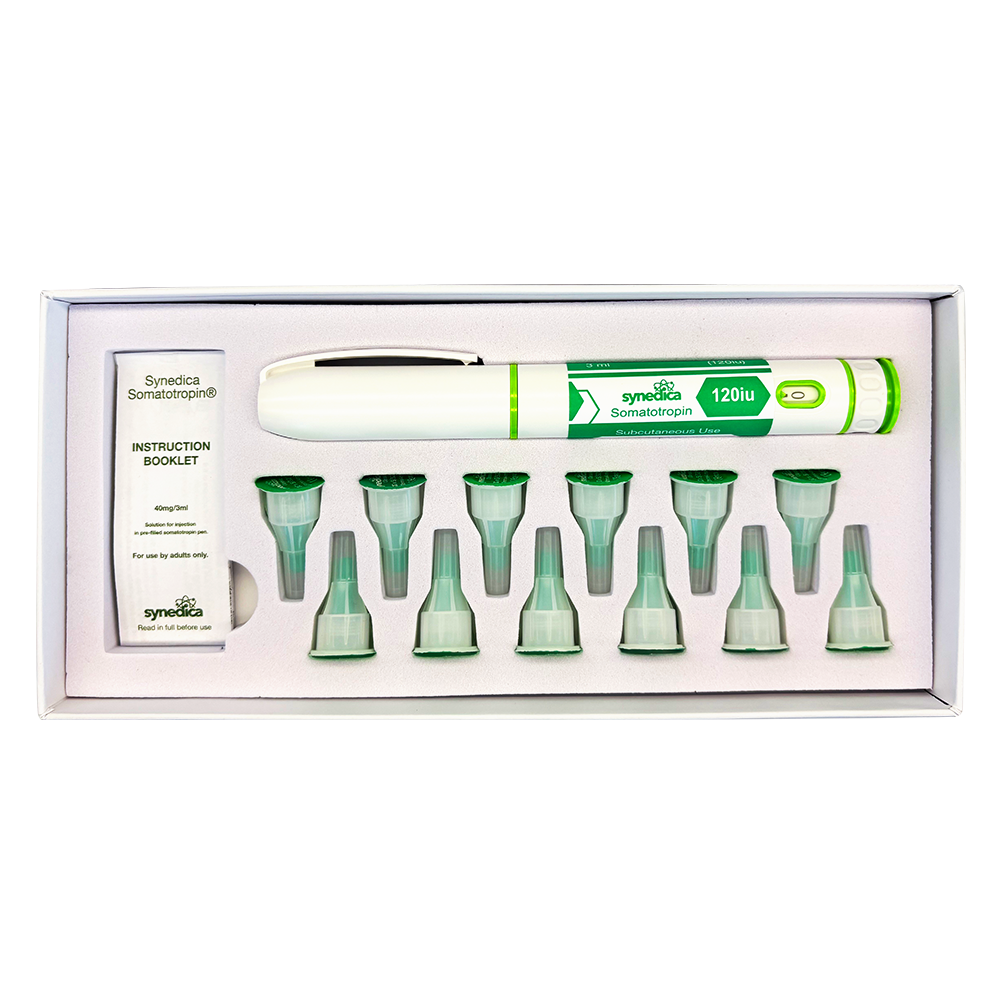
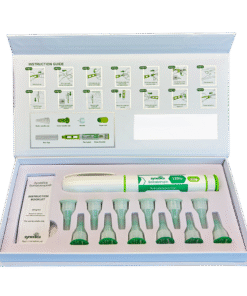
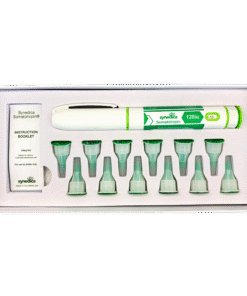
The Synedica TRT + HCG Daily Injection Pen Kit provides a 14-day research supply and includes one precision dosing injection pen, fourteen sterile disposable needles, and a detailed instruction booklet for laboratory handling. Each measured dose contains 15mg of testosterone cypionate and 150 IU of HCG, allowing for consistent and repeatable administration within controlled study protocols.
The micro-dosing format is specifically designed for research into steady-state hormone levels and daily administration models. Within laboratory environments, this approach enables detailed observation of hormonal fluctuations, receptor activity, and metabolic response under consistent dosing conditions. The subcutaneous delivery method further supports studies into absorption rates and administration efficiency.
Across the UK and Europe, testosterone and HCG-based compounds are subject to strict regulation and are typically only available for medical use via prescription. In countries such as Germany, access may be provided through licensed healthcare providers or covered under statutory health insurance for eligible individuals, depending on clinical requirements.







Reviews
There are no reviews yet.